When Classical Physics Stops Working
Classical mechanics tells us that everything in the universe follows clear and predictable rules. If we know the initial conditions, external influences, and how objects interact, we can calculate their motion with remarkable precision.
From planetary orbits to GPS tracking on a smartphone, this framework works extremely well. If an object is standing still, its position is fixed and its velocity is simply zero.
However, this certainty begins to break down when we move into extreme conditions. At very small scales, such as atoms, or at velocities approaching the speed of light, nature no longer follows the same rules. Instead, entirely different laws take over.
A Particle Is Not a Point
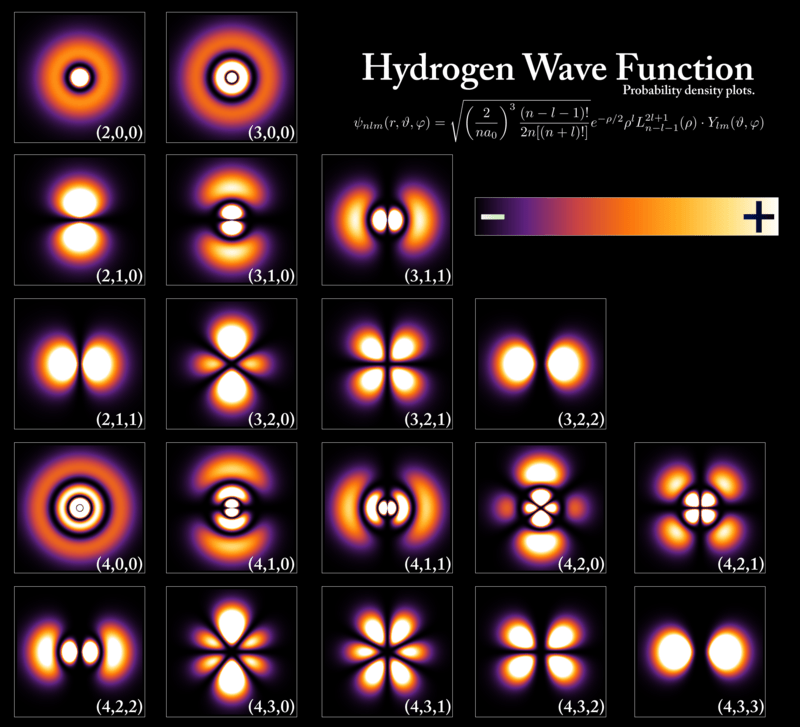
In quantum mechanics, a particle does not behave like a tiny solid object sitting at a fixed position. Instead, its existence is described by probabilities.
Rather than having a single exact location, a particle can exist across many possible positions at once, each associated with a certain probability of being observed there.
This behavior is governed by the wave function, a mathematical description of how a particle evolves over time. What we perceive as a “position” is simply where the particle is most likely to be detected, not where it definitively exists.
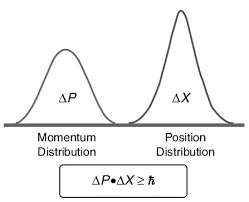
The Trade-Off Between Position and Velocity
To measure the position of something like an electron, we need to interact with it. One common idea is to use photons to detect its location.
The problem is that photons carry energy. According to quantum theory, the energy of a photon is proportional to its frequency. High-frequency photons carry more energy, while low-frequency photons carry less.
If we use high-energy photons, we can pinpoint the electron’s position more precisely. But in doing so, we disturb the electron, changing its velocity. If we use low-energy photons, we disturb it less, but our measurement becomes less precise.
This creates an unavoidable trade-off. The more precisely we know a particle’s position, the less precisely we can know its velocity, and vice versa. This is not a limitation of technology, but a fundamental property of nature.
Trapping an Electron: A Thought Experiment
Imagine placing an electron inside a perfectly sealed box and gradually shrinking the box to confine it into a smaller space.
At first glance, it seems reasonable to expect that by shrinking the box enough, we could force the electron into a fixed position with zero velocity.
But something unexpected happens. As the box becomes smaller, the electron’s motion becomes increasingly erratic. Its energy rises, and its velocity becomes more unpredictable.
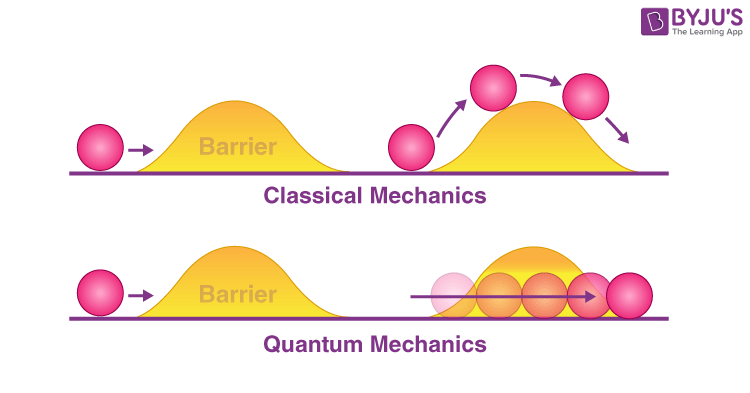
If we continue shrinking the box, the electron may even escape entirely through a process known as quantum tunneling. It does not break the walls or find a gap—it simply appears outside.
This thought experiment reveals a deep truth: particles cannot be confined arbitrarily without consequences. The act of confinement itself increases their energy.
Where Does the Energy Come From?
This leads to a natural question. If the box is empty, where does the extra energy come from?
In quantum physics, empty space is not truly empty. It contains fluctuations—temporary changes in energy that appear and disappear over extremely short timescales.
A useful way to imagine this is through a simplified analogy. When a system is forced into an extreme condition, it can “borrow” energy from the vacuum, as long as that energy is returned quickly enough.
This idea is closely related to another form of uncertainty: the relationship between energy and time. The shorter the duration of an event, the greater the uncertainty in its energy.
In other words, for very brief moments, nature allows energy to fluctuate beyond what we would normally expect.
The Energy–Time Uncertainty
Beyond position and velocity, quantum mechanics also reveals a second fundamental limitation: the relationship between energy and time.
If we measure the duration of an event very precisely, the total energy involved becomes uncertain. Conversely, if we measure energy precisely, we lose accuracy in timing.
Short-lived events can involve large energy fluctuations, while long-lasting processes tend to stabilize at lower energy levels.
This principle helps explain how particles can briefly access energy that does not seem to exist in classical terms, as long as it is returned within an extremely short time.
The Deep Meaning of Uncertainty
The uncertainty principle is not just a technical detail of quantum mechanics. It is one of its central foundations.
Concepts that seem obvious in everyday life—such as having a definite position and velocity—do not apply in the same way at the quantum level.
Instead, reality at small scales is inherently probabilistic. The limitations we encounter are not due to imperfect instruments, but are built into the structure of nature itself.
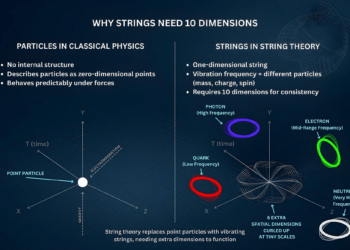
When these ideas are extended to spacetime and fundamental physics, they reveal deeper inconsistencies that remain unresolved to this day. These unresolved tensions are at the heart of some of the biggest problems in modern physics.