The Chemical History Written in the Milky Way
Spectroscopic observations show that the ten most common elements in the Milky Way include hydrogen, helium, oxygen, carbon, neon, iron, nitrogen, silicon, magnesium, and sulfur. This chemical composition reveals something important about our galaxy’s past. It tells us that the Milky Way has experienced a long history of stellar birth, evolution, and death.
Many of the stars that contributed to this chemical mixture were relatively small or medium-sized stars. According to stellar evolution models, stars of this type typically live for billions—or even tens of billions—of years. The abundance of elements such as oxygen, carbon, and iron indicates that numerous stellar explosions have already occurred throughout the galaxy’s history.
These events include phenomena like novae and Type Ia supernovae, which disperse newly formed elements into interstellar space.
The Sun and the Second Generation of Stars
Today the Sun is believed to be a second-generation star. In other words, it formed from interstellar clouds that were enriched by earlier generations of stars that had already exploded.

These clouds contained heavier chemical elements—often called “metals” in astrophysics—which were created inside previous stars. The presence of these heavier elements is what allowed planets such as Earth to form.
Our planet itself is composed of many complex elements. The most abundant include oxygen, silicon, aluminum, iron, calcium, sodium, magnesium, potassium, titanium, and hydrogen. Although hydrogen atoms are extremely numerous, the ranking of these elements usually refers to their contribution by mass.
Earth also contains trace amounts of much heavier elements such as gold, platinum, and lead.
The First Stars of the Universe
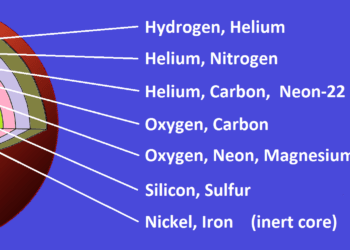
The star that existed before the formation of our Solar System must have been extremely massive. Only very large stars can produce elements heavier than iron through the violent processes that occur during supernova explosions.

These ancient giants belonged to what astronomers call the first generation of stars, sometimes referred to as primordial stars. Such stars formed from almost perfectly pure hydrogen and helium, the only elements produced shortly after the Big Bang.
Primordial stars were enormous—often hundreds or even thousands of times more massive than the Sun—and they lived very short lives. Because of this, none of them are expected to exist today.
Why a Pure Universe Could Not Create Life
In modern astrophysics, primordial stars remain largely theoretical. They likely appeared shortly after the Big Bang and quickly disappeared after exploding as supernovae. Their explosions seeded the universe with heavier elements.
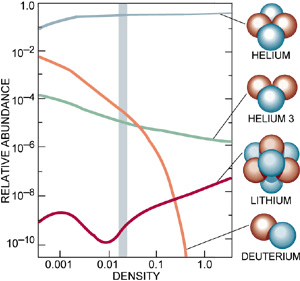
This process had profound consequences. The early universe, composed almost entirely of hydrogen and helium, was chemically simple. Without heavier elements such as carbon, oxygen, nitrogen, or iron, the formation of planets and complex chemistry would have been extremely unlikely.
In other words, a perfectly pure universe would have been a universe without life.
The Chemical Ingredients of Living Systems
When we look at Earth from space, its surface appears to be dominated by oceans and continents. Chemically speaking, oxygen and silicon are among the most common elements found in rocks, minerals, and planetary crusts.
Yet the most remarkable feature of our planet is not its geology—it is life itself.
Life on Earth is extraordinarily diverse. In fact, scientists estimate that most forms of life are microscopic organisms that remain largely unknown to us. Despite this diversity, nearly all life shares a common molecular foundation: DNA.
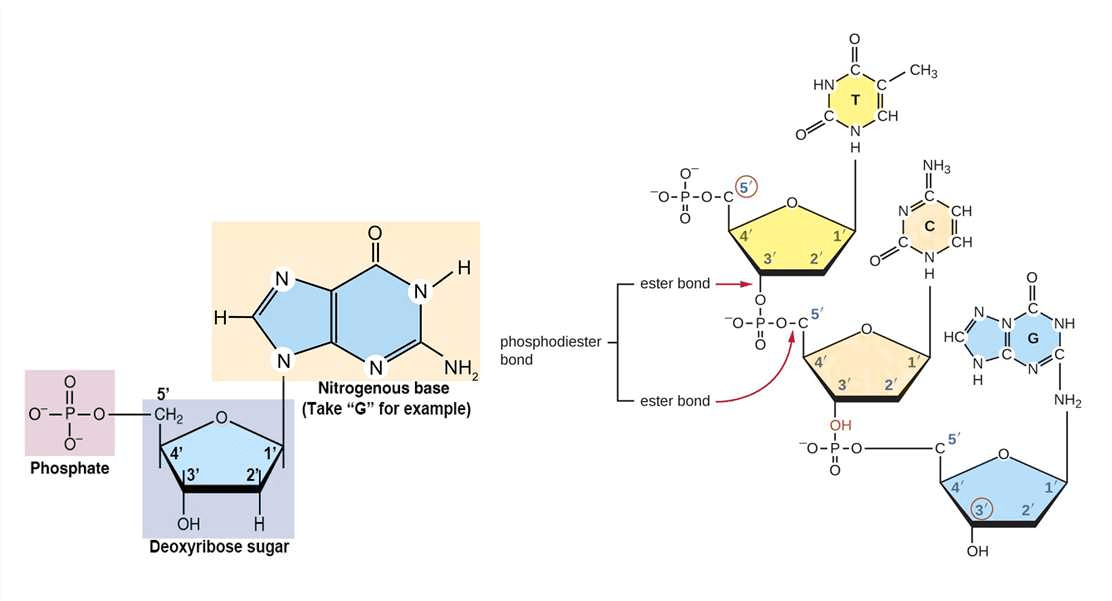
DNA stores genetic information and controls the synthesis of proteins within living cells. At its core lies carbon chemistry. Carbon atoms form the structural backbone of DNA molecules, together with oxygen, nitrogen, and hydrogen.
Unlike hydrogen and helium, carbon was not created during the Big Bang. It formed later inside stars and was released into space when those stars died.
We Are Made of Stellar Debris
Even the human body reflects this cosmic origin. By mass, the average human body contains roughly 65% oxygen, 18% carbon, nearly 10% hydrogen, about 3% nitrogen, and smaller amounts of calcium, phosphorus, potassium, sodium, chlorine, magnesium, iron, zinc, and other elements.

With the exception of hydrogen, nearly all of these elements were created inside stars or during supernova explosions. In a very real sense, the atoms that make up living organisms were forged in stellar furnaces long before the Solar System existed.
Stars therefore act as cosmic factories, producing the chemical ingredients required for life.
A Universe Ready for Life
When we examine the chemical composition of the Milky Way today, it becomes clear that the ingredients for life are now widespread. The universe has evolved from a simple primordial mixture into a chemically rich environment.

Although life may theoretically have appeared after the first generations of stars exploded, the early universe was an extremely chaotic place. Violent cosmic events were common, making stable planetary environments rare.
Only after billions of years—when many generations of stars had lived and died—did the universe become chemically mature enough to support planetary systems like our own.
In that sense, the current cosmic era may represent one of the best times for life to emerge in the history of the universe.