A Particle That Never Experiences Time
According to modern physics, the life of a photon is strangely simple. The moment a photon is created, its journey toward destruction has already begun. In the four-dimensional spacetime described by Albert Einstein, a photon travels at the speed of light, and this has an extraordinary consequence.
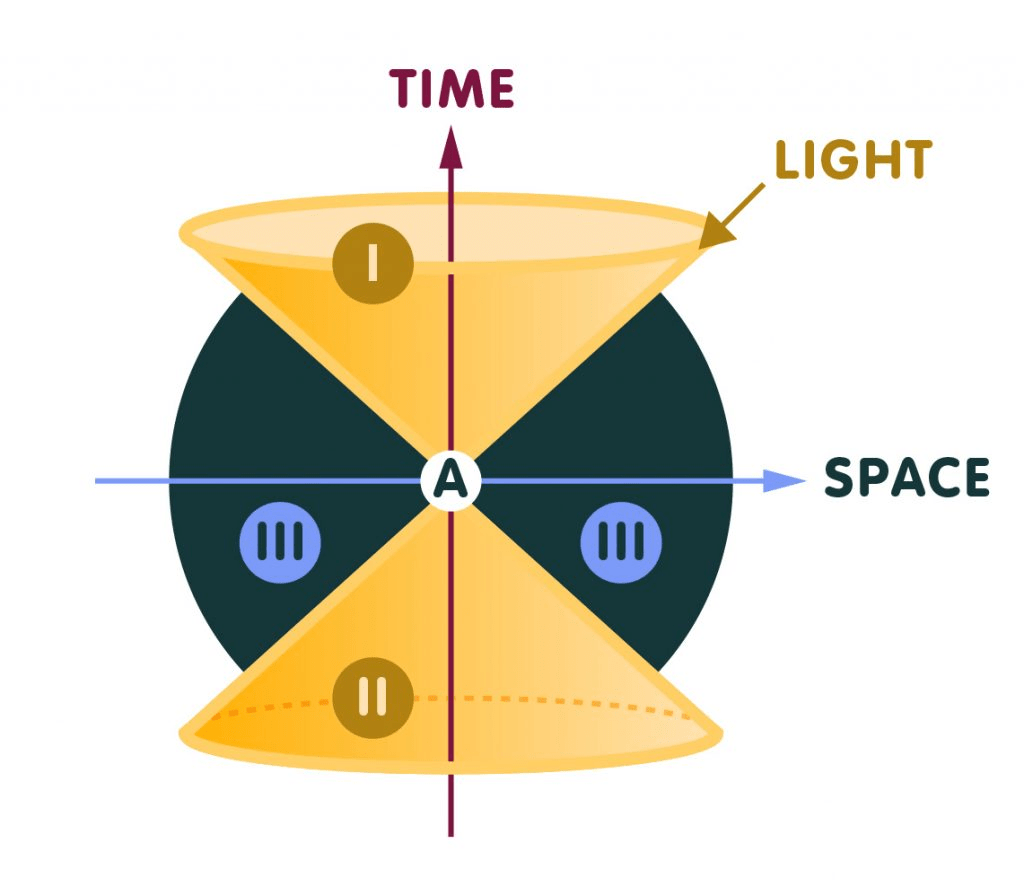
From the photon’s own perspective, time does not pass.
Because it moves at light speed, the photon experiences no passage of time between its creation and its final interaction. In a sense, the entire journey of a photon—whether it travels a meter or billions of light-years—occurs instantaneously from its own frame of reference. Its “life” consists of only two moments: the instant it is emitted and the instant it interacts with something else.
This strange idea makes photons among the most unusual particles in the universe.
Are Photons Truly Stable Particles?
With our current understanding of particle physics, photons are considered extremely stable. If photons were not stable, the universe would look very different. Light emitted by distant galaxies would decay long before reaching us, and the cosmos would appear far darker than it does today.
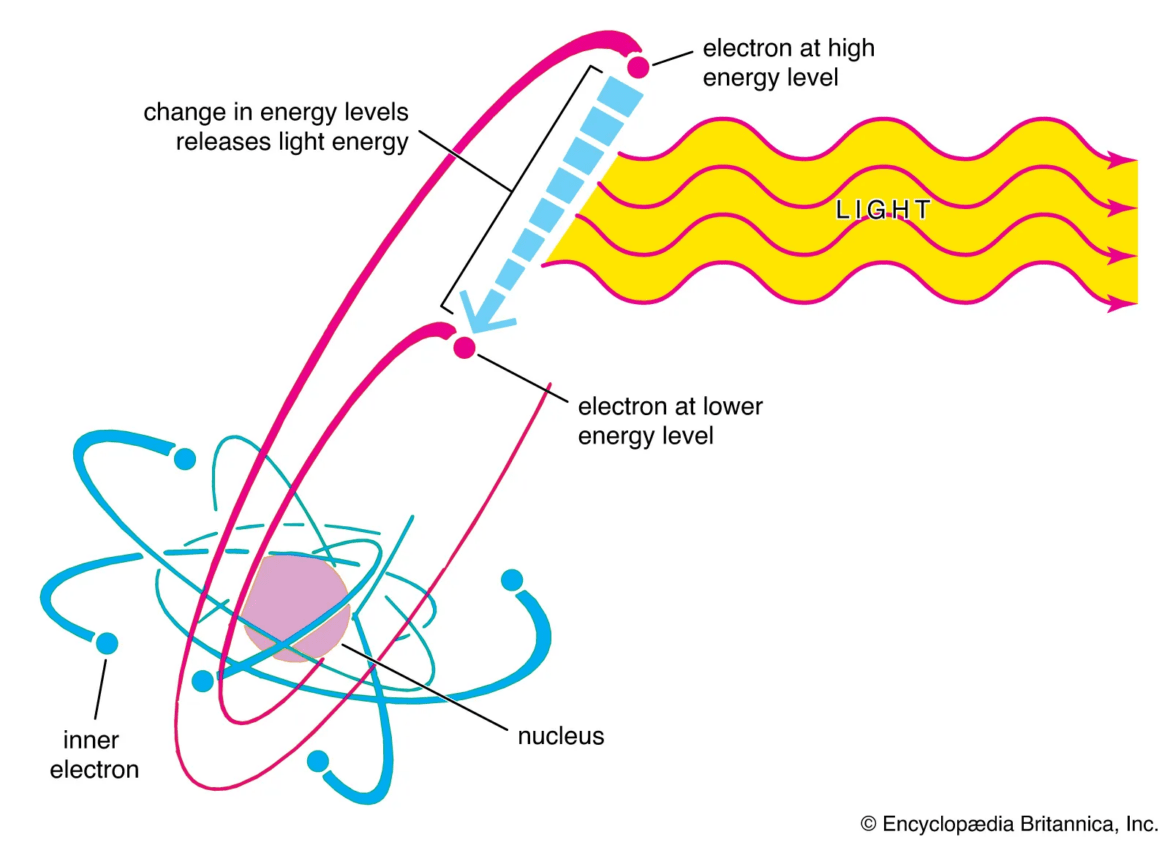
In modern particle physics, several particles are believed to be fundamentally stable under normal conditions. These include the photon, the electron, and the gluon. Another particle often considered extremely long-lived is the proton.
Although proton decay has been predicted by some theories, it has never been observed. Experimental limits suggest that a proton could survive for at least 10³⁰ to 10³⁶ years. If protons were unstable on shorter timescales, the structure of matter itself would eventually collapse.
Fortunately for life, matter appears to be remarkably durable.
When Photons Transform Instead of Decay
Even though photons do not decay in the usual sense, they can still participate in unusual physical processes. Under extreme conditions, a photon may interact with other particles or fields in ways that transform its energy.
For example, two very energetic photons can collide and produce a pair of particles and antiparticles. This process converts energy directly into mass, illustrating one of the most famous consequences of relativity.
A high-energy photon may also interact with an electron and lose a large fraction of its energy, becoming a lower-energy photon. In certain nonlinear media, a photon can even split into multiple photons with lower energies.
These events are rare and require special environments, but they demonstrate that photons can change form without truly disappearing.
Cosmic Expansion and the Loss of Photon Energy
One of the most important large-scale effects acting on photons comes from the expansion of the universe. In the 1920s, Edwin Hubble showed that distant galaxies are moving away from us, revealing that the universe itself is expanding.
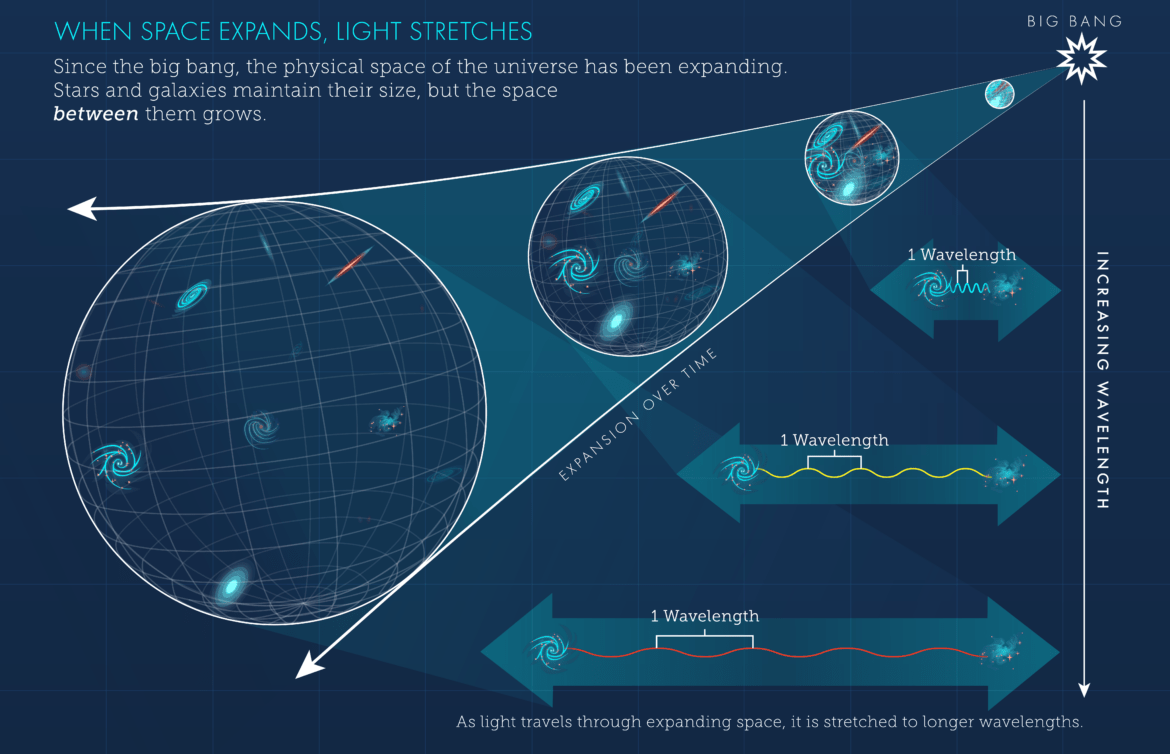
As space expands, the wavelength of traveling light stretches along with it. This phenomenon is known as redshift. The speed of a photon does not change—it always travels at the speed of light—but its wavelength becomes longer and its energy decreases.
Over extremely long cosmic timescales, photons traveling across expanding space gradually lose energy. Their frequencies drop lower and lower, shifting from visible light to infrared, then microwave, and eventually to extremely weak radiation.
The Idea of the Heat Death of the Universe
If cosmic expansion continues indefinitely, physicists predict a distant future known as the heat death of the universe. In this scenario, stars eventually burn out, galaxies grow dark, and only faint radiation remains drifting through space.

The remaining photons would become so stretched and diluted that they would barely interact with anything. The universe would become extremely cold and dark.
Temperatures would approach absolute zero, and the cosmos would slowly drift toward a state where no significant energy differences remain.
Why Absolute Zero May Never Be Reached
Despite this bleak prediction, physics suggests that the universe may never actually reach perfect absolute zero. Several fundamental principles prevent complete stillness at the quantum level.
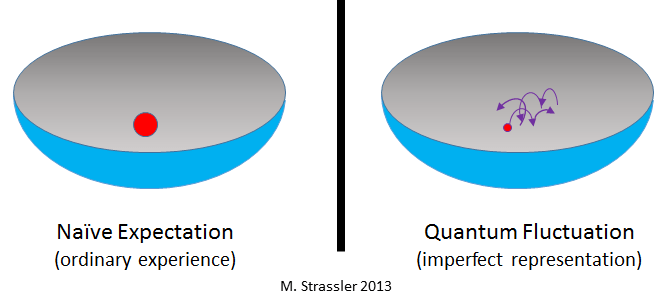
One important concept is the Heisenberg Uncertainty Principle, which states that the position and momentum of a particle cannot both be known with perfect precision. This uncertainty implies that particles can never be completely motionless.
Another principle is the Pauli Exclusion Principle. It prevents identical fermions—such as electrons—from occupying the same quantum state simultaneously. This rule ensures that electrons maintain distinct energy levels even at extremely low temperatures.
Finally, quantum fluctuations guarantee that even the emptiest vacuum still contains a small amount of energy.
Because of these effects, matter and radiation can never become perfectly static. Even in the coldest conceivable future of the universe, the quantum world will continue to vibrate with tiny fluctuations of energy.