Energy at the Beginning of the Universe
Around 14 billion years ago the universe began with an event that released an unimaginable amount of energy. Although scientists are still refining the details of that moment, commonly described by the Big Bang theory, one idea is widely accepted: everything that exists today is ultimately a form of energy.

According to the law of conservation of energy, energy cannot simply disappear. Instead, it changes form. A small portion of the energy present in the early universe eventually condensed into matter. Today, ordinary matter — the atoms that form stars, planets, and living organisms — makes up only about five percent of the total content of the cosmos.
This means every atom around us is essentially a compressed storage unit of energy. Just like a hard drive stores information in a tiny space, atoms store enormous amounts of energy inside an extremely small structure.
Atoms and the Structure of Matter
Everything around us is built from atoms. Your body, the air, the Earth, and even the Sun are made of these tiny structures. But atoms themselves are not fundamental — they are composed of smaller particles.
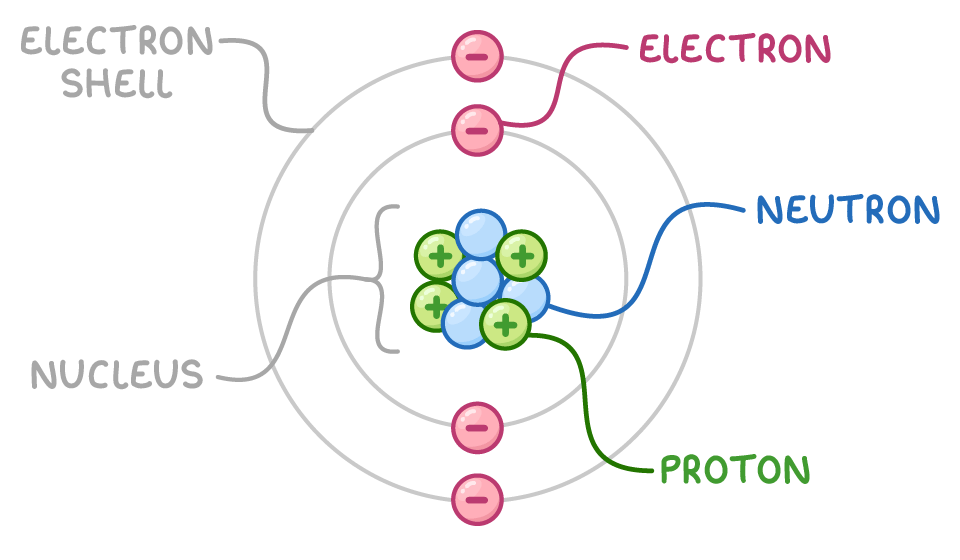
Among the most important components are quarks, electrons, gluons, and photons. These particles interact through fundamental forces and form the structures that make up atomic nuclei and electron clouds.
The stability of matter depends on these interactions. When particles bind together through fundamental forces, energy is stored inside those bonds. The stronger the bond, the more energy is trapped inside the structure.
The Role of Electrons
Electrons are among the most familiar particles in physics. Despite their extremely small mass, they play a central role in chemistry, electricity, and modern technology.
Inside atoms, electrons move around the nucleus and interact through electromagnetic forces. These interactions are mediated by photons, which carry the electromagnetic force between charged particles.
When humanity discovered how to control the movement of electrons — electric current — civilization entered a new technological era. Nearly all modern technology relies on the controlled flow of electrons.
However, despite their importance, electrons store only a tiny fraction of an atom’s total energy.
Mass and Energy
The relationship between mass and energy was famously described by Albert Einstein with the equation:
E = mc²
This equation reveals that mass itself is a form of energy. Even a tiny amount of mass can correspond to an enormous amount of energy when converted.
Although electrons contribute a little to the total energy of an atom, their mass is extremely small. Most of the energy stored in matter comes from a different place entirely — the atomic nucleus.
The Energy Inside the Atomic Nucleus
At the center of every atom lies its nucleus, made of protons and neutrons. These particles are themselves built from smaller components called quarks, which are held together by particles known as gluons.
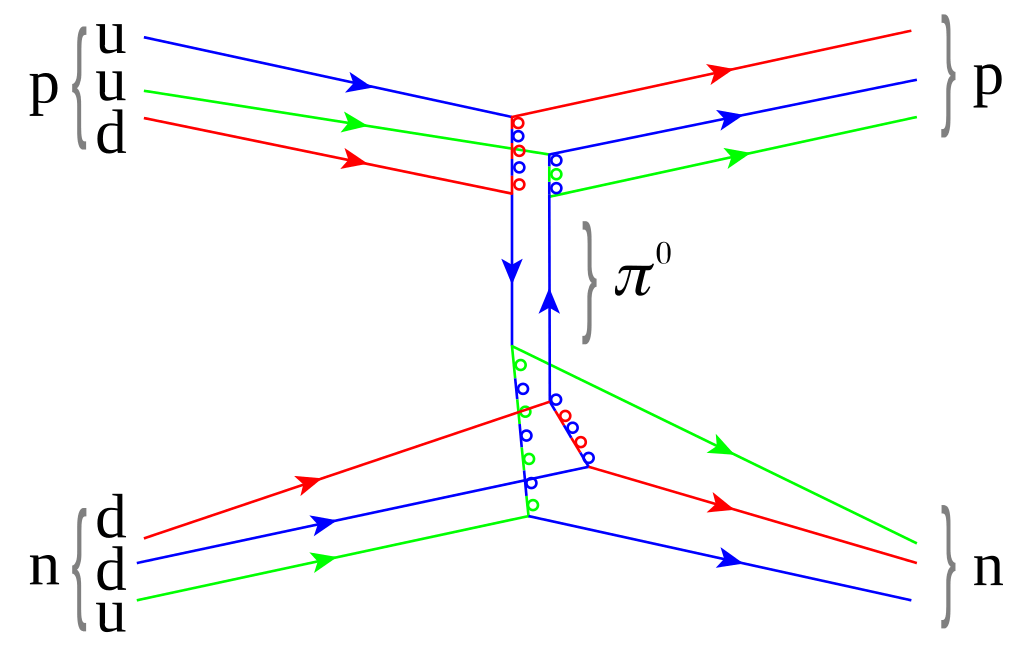
Gluons carry the strong nuclear force, the most powerful force in nature. This force binds quarks together and holds the nucleus intact despite the electrical repulsion between protons.
In fact, almost 99 percent of the mass of an atom arises from the energy associated with the strong nuclear force. This means nearly all of the energy contained in matter is stored within the nucleus.
Breaking the Atom
Atoms are extremely stable structures. Removing electrons is relatively easy, but breaking apart the nucleus is far more difficult.

Modern science cannot completely destroy an atom in a practical way, but it can release part of the energy stored inside the nucleus. One method is nuclear fission, where the nucleus of a heavy atom splits into smaller pieces.
A famous example is uranium-235. When its nucleus absorbs a neutron, it becomes unstable and splits into smaller atoms while releasing energy and additional neutrons. These neutrons can trigger further reactions, creating a chain reaction.
The energy released during this process comes from the breaking of strong nuclear bonds inside the nucleus.
The True Scale of Atomic Energy
Even nuclear fission only releases a small fraction of the total energy contained within matter. According to Einstein’s equation, converting the entire mass of an atom directly into energy would release far more power.
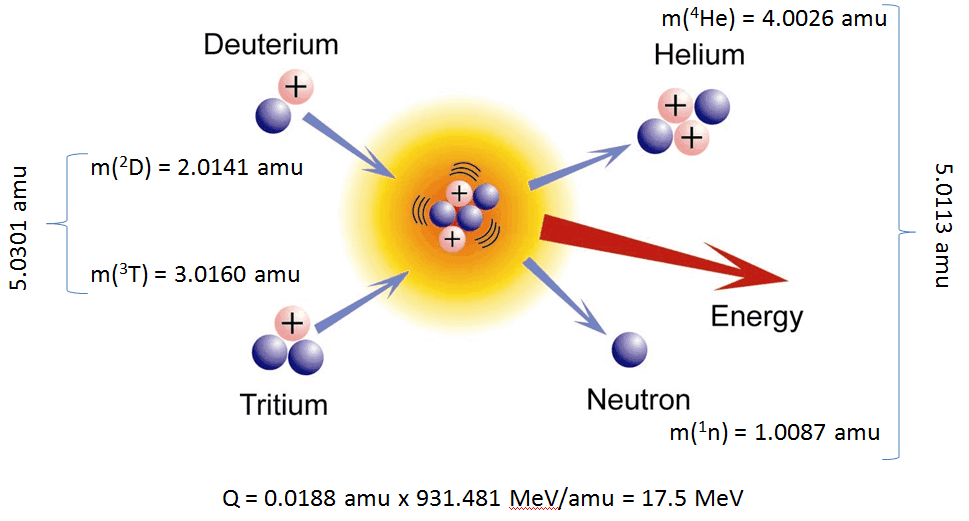
This leads to a remarkable conclusion: every atom in the universe contains an enormous reservoir of energy, compressed into one of the smallest structures in nature.