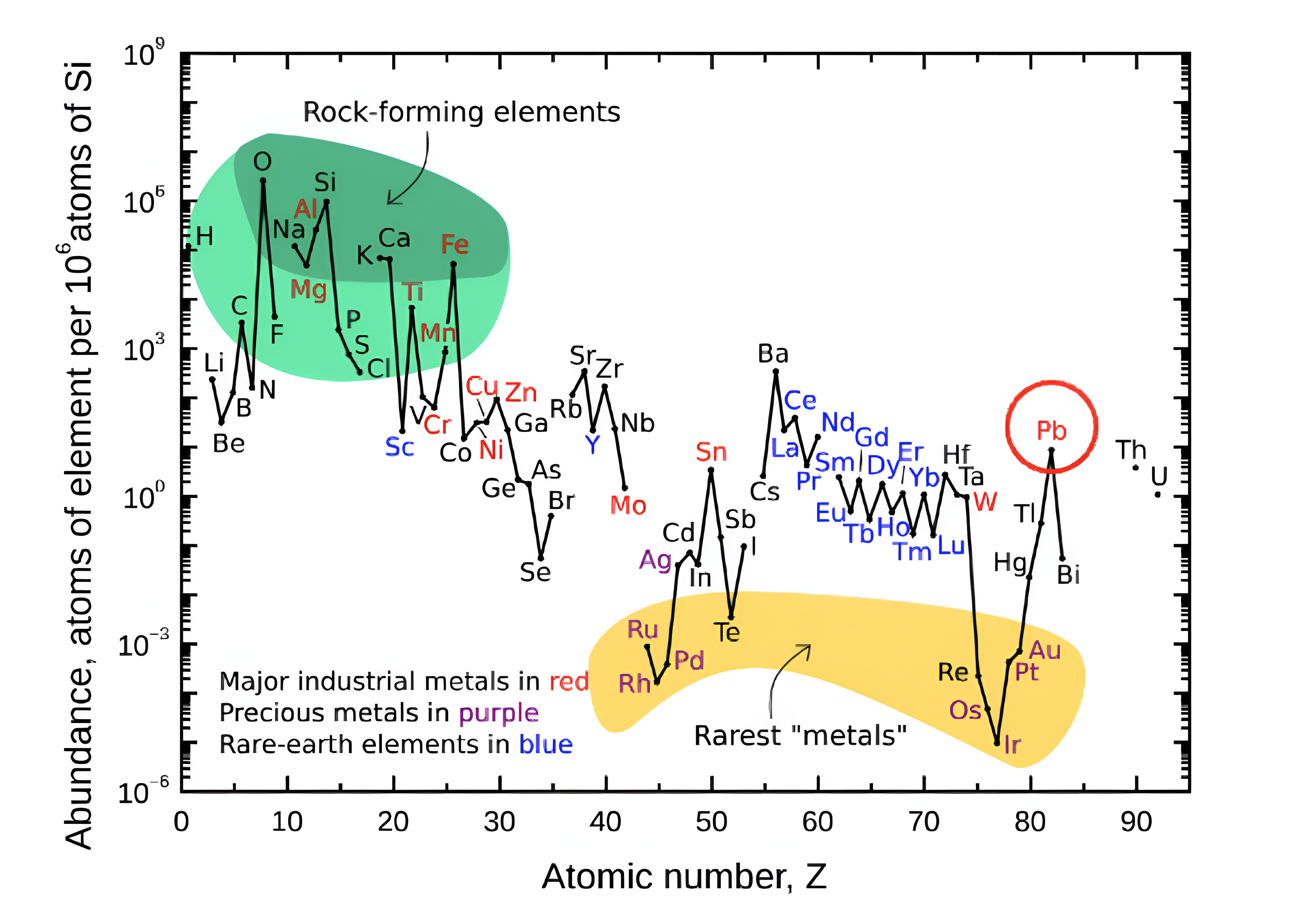
Stars Are the Cosmic Factories of Matter
Every star shining in the night sky can be imagined as a giant cosmic furnace. Deep inside their cores, stars release enormous energy through nuclear fusion. In this process, light elements gradually combine to form heavier ones.
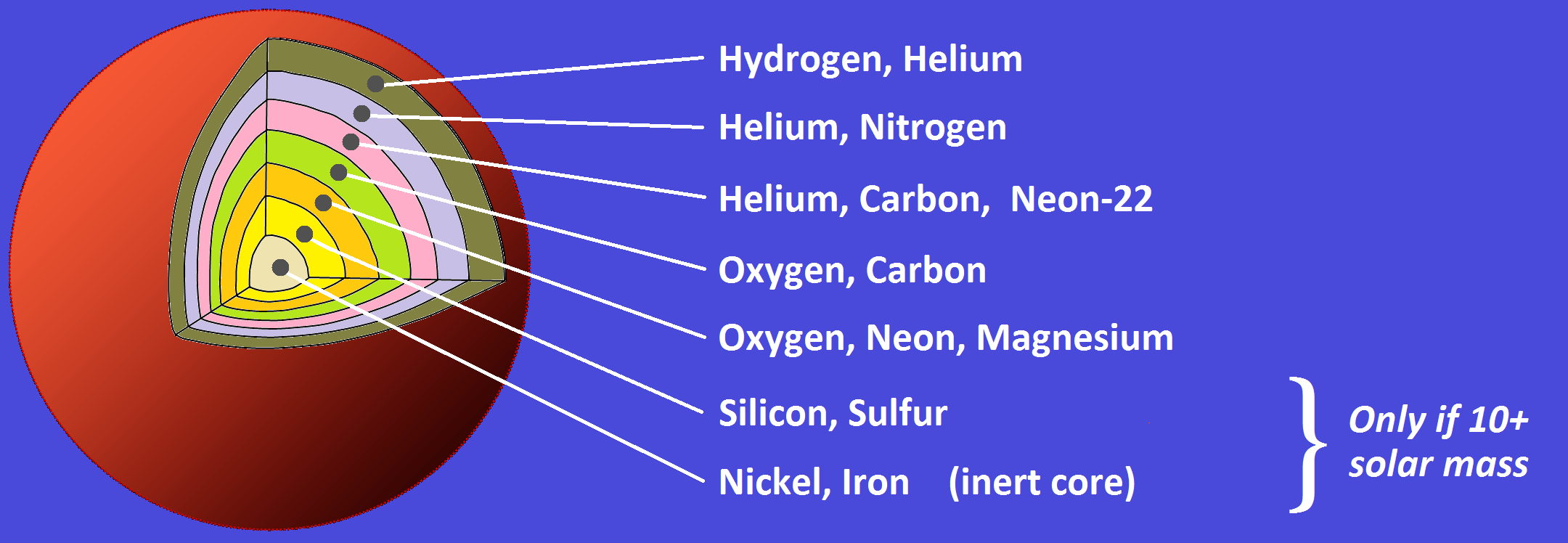
At the beginning of a star’s life, hydrogen nuclei fuse together to form helium. As temperatures rise inside the stellar core, more complex reactions begin to occur. Carbon, oxygen, neon, and silicon are formed step by step through successive fusion stages. Over millions and billions of years, stars become the primary factories that manufacture most of the elements found throughout the universe.
But this cosmic production line eventually reaches a surprising limit.
Iron: The Boundary Where Fusion Stops Releasing Energy
One of the most important turning points in stellar evolution occurs when a star begins producing iron in its core. Iron occupies a special position in nuclear physics and marks the boundary of energy-producing fusion inside stars.

4
Fusion reactions involving elements lighter than iron release energy. The mass difference between the original particles and the final nucleus is converted into energy, helping the star resist gravitational collapse.
Iron behaves differently. When nuclei heavier than iron attempt to fuse, the reaction actually consumes energy rather than releasing it. Once large quantities of iron accumulate in the core, the delicate balance that keeps the star stable begins to break down.
The Hidden Forces Inside Atomic Nuclei
The reason iron forms this boundary lies in the balance between two fundamental forces inside atomic nuclei.

The first is the strong nuclear force, the most powerful force in nature at extremely short distances. It binds protons and neutrons together inside the nucleus.
Opposing it is the electromagnetic force. Because protons carry positive electric charge, they repel one another. As more protons accumulate in a nucleus, this repulsive force becomes stronger. Eventually the stability of the nucleus begins to weaken, which is why extremely heavy elements are often unstable and radioactive.
Slow Neutron Capture in Massive Stars
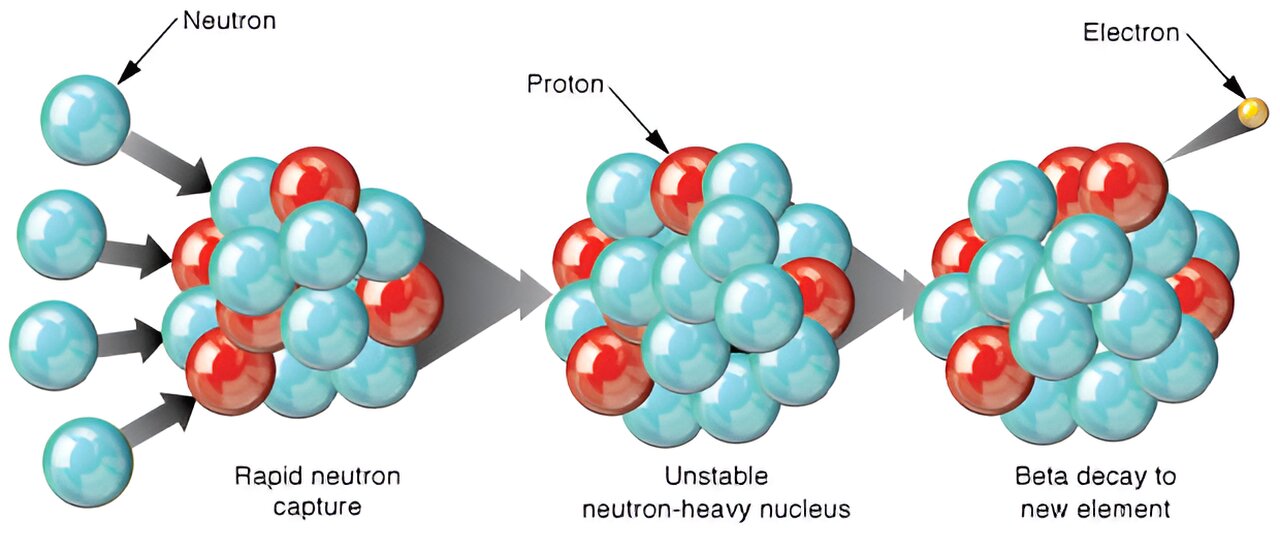
Even though ordinary fusion stops at iron, stars still possess another mechanism capable of producing heavier elements. This process is known as slow neutron capture.
In the late stages of stellar evolution, particularly in massive stars, nuclear reactions can release free neutrons. These neutrons may collide with heavy nuclei and become absorbed.
When a nucleus captures a neutron, it can become unstable. Through beta decay, the neutron may transform into a proton, turning the atom into a new element with a higher atomic number. Over time this process slowly builds heavier elements inside aging stars.
However, this mechanism also has a natural limit.
When Neutron Capture Must Become Rapid
The slow neutron capture process eventually reaches a barrier near the element bismuth. Beyond this point, unstable nuclei decay too quickly to continue absorbing additional neutrons.
To form even heavier elements, neutron capture must occur extremely rapidly. Instead of waiting for radioactive decay, nuclei must absorb large numbers of neutrons in a very short time.
This mechanism is known as rapid neutron capture, often called the r-process. It requires environments filled with enormous numbers of energetic neutrons, conditions far more extreme than those found inside ordinary stars.
For decades, astronomers searched for cosmic environments where such conditions might exist.
Neutron Star Collisions Forge the Heaviest Elements
One of the most powerful environments capable of producing the r-process occurs during collisions between neutron stars.

Neutron stars are incredibly dense stellar remnants left behind after massive stars explode as supernovae. When two of these objects spiral together and collide, they eject vast amounts of neutron-rich material into space.
In 2017, gravitational-wave detectors recorded the merger of two neutron stars in a galaxy about 140 million light-years away. Telescopes around the world soon observed the explosion that followed and detected the spectral signatures of heavy elements such as gold, uranium, and platinum.
These observations strongly suggest that many of the heavy metals found on Earth were forged during such cosmic collisions long before the Solar System was born. In other words, some of the rarest elements on our planet were created in the most violent events the universe can produce.